Author: Emmanuel Oyelayo

Background
Imagine seeking help for a painful urinary tract infection (UTI). The doctor, constrained by time and technology, prescribes a broad-spectrum antibiotic, hoping it works. However, without knowing the specific bacteria or its resistances, this is an educated guess. You leave the clinic with a prescription, but the uncertainty about “Will this drug actually work?” lingers.
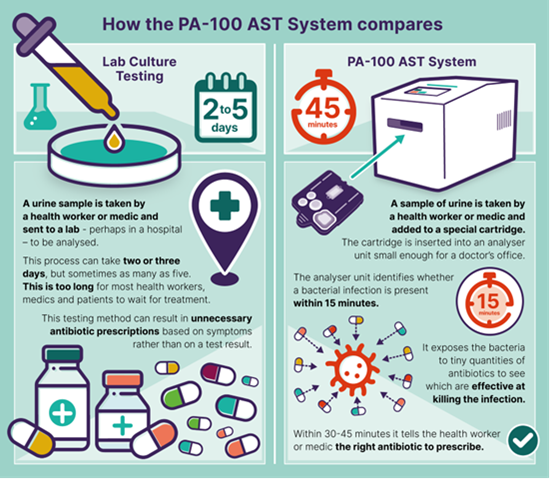
This scenario is the reality for millions globally. For decades, the gold standard for diagnosing bacterial infections and determining effective antibiotics has relied on culturing bacteria. While accurate, this process takes 2–3 days [1]. During this critical window, healthcare providers are forced into empirical prescribing (administering antibiotics based on experience rather than specific evidence) [2]
This “wait and guess” approach has dire consequences, whereby patients may suffer treatment failure and side effects from unnecessary drugs. More critically, it fuels the engine of Antimicrobial Resistance (AMR), where bacteria learn to outsmart the very drugs designed to kill them. The World Health Organization has identified AMR as one of the top global public health threats, driven significantly by the misuse and overuse of antibiotics [3].
Urinary tract infections (UTIs), one of the most common bacterial infections worldwide with over 400 million cases in 2019, are a prime example of this crisis [4]. The gap between symptom onset and effective treatment is exactly the void the PA-100 AST System was designed to fill.
The Solution: Introducing the PA-100 AST System
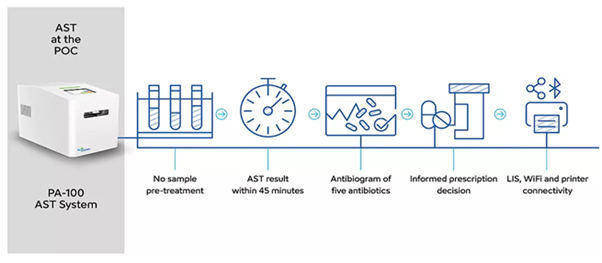
Developed by Sysmex Corporation and Sysmex Astrego, the PA-100 AST represents a paradigm shift in diagnostic medicine. It is a rapid, point-of-care diagnostic device that fundamentally changes the clinical workflow.
The system provides two critical answers within a single doctor’s visit which includes; whether bacteria are present and, crucially, which specific antibiotic will kill them [5].

How It Works: The Science Simplified
Unlike conventional methods that wait for bacteria to grow in a lab over days, the PA-100 uses advanced nanofluidic technology combined with phase-contrast microscopy to observe bacteria at a single-cell level [6].
The process is elegantly simple:
- A fresh urine sample (just 400 microliters) is pipetted into a smartphone-sized disposable cartridge [7].
- The cartridge contains a chip with over 11,000 individual nanochannels. Bacteria are trapped in these channels, isolating them for observation [6].
- The trapped bacteria are exposed to five different antibiotics (e.g., nitrofurantoin, fosfomycin, trimethoprim, amoxicillin/clavulanate, and ciprofloxacin) that align with European and North American guidelines for uncomplicated UTIs (EUCAST breakpoints). A high-resolution microscope continuously monitors their growth.
- Because the system watches the bacteria grow (or die) in real-time, it produces a phenotypic antibiotic susceptibility test (AST) result, which is the same “gold standard” used by central labs, but in minutes, not days [4].
Speed and Capabilities
- Results out in 30–45 minutes total [5].
- Currently validated for the five most common uncomplicated UTI pathogens: Escherichia coli, Klebsiella pneumoniae, Proteus mirabilis, Enterococcus faecalis, and Staphylococcus saprophyticus [4].
Scientific Validation
Innovation is meaningless without rigorous validation. The PA-100 has undergone significant clinical scrutiny. A landmark prospective study published in the European Journal of Clinical Microbiology & Infectious Diseases in 2024 evaluated the system on 278 female patients with suspected uncomplicated UTIs [4].
The findings were striking:
- The system demonstrated a 99.4% specificity in detecting bacterial infections, meaning false positives are extremely rare [4].
- The PA-100 provided an optimal treatment recommendation in 78.4% of cases. In comparison, unaided clinical judgement (empirical prescribing) achieved only a 58.3% accuracy rate [4].
- Since it accurately identifies effective antibiotics, the system facilitates the potential return of “retired” first-line antibiotics, reducing our reliance on broad-spectrum last-resort drugs [8].
Global Recognition: Winning the Longitude Prize on AMR
The most definitive external validation of the PA-100’s potential came in June 2024, when it was awarded the £8 million Longitude Prize on AMR [9]. This prestigious prize, launched by Challenge Works and Nesta, was designed to incentivize the creation of an accurate, rapid, and easy-to-use test for bacterial infections.
Selected from over 250 global competitors, the PA-100 was lauded for its ability to “bring the power of laboratory testing into a doctor’s office” [7]. Dame Sally Davies, the UK Special Envoy on AMR, noted that such technology is critical to ending empirical prescribing, which has fueled the resistance crisis [7].

Relevance to Africa and Nigeria
While the PA-100 is currently in early adoption phases in Europe, its implications for African healthcare systems, including Nigeria, are profound. The manufacturers, Sysmex, have concrete expansion plans for the continent. In 2025, the company established a new subsidiary in Nairobi, Kenya, as its fifth base in Africa, and has deepened its collaboration with partners in Nigeria to introduce new diagnostic tools.
Across much of Africa, access to functional microbiology laboratories is severely limited. In primary care centers and rural clinics, UTIs and other infections are treated entirely empirically. This high-resistance, low-diagnostic environment is a breeding ground for superbugs.
The Potential Impact:
If deployed strategically, the PA-100 could revolutionise infection management in several ways:
- The device is maintenance-free and requires no specialist microbiological training, making it viable for rural clinics and primary health centers where standard lab infrastructure does not exist [4].
- In many African nations, access to newer antibiotics is limited. By ensuring that cheap, first-line antibiotics (like nitrofurantoin or trimethoprim) are effective before prescription, the PA-100 can preserve the efficacy of the limited formulary.
- It provides a tangible tool for hospitals to implement stewardship programs that can influence policy to digital, evidence-based action at the bedside.
- In Nigeria and some other African countries, nitrofurantoin is not routinely prescribed due to higher resistance rates and different local treatment algorithms (e.g., cefalexin, fluoroquinolones, or co-amoxiclav are often preferred). Therefore, the device’s current cartridge would require region-specific recalibration before it can deliver clinically meaningful results in Nigeria and some other African countries.
Current Status and Implementation Challenges
As of late 2024 and into 2025, the system has received its CE (Conformité Européenne which is French for “European Conformity”) mark and market introduction has commenced in Europe [8]. Ongoing evaluations, such as the TOUCAN (plaTform fOr Urinary tract infection diagnostiC evAluatioN) study with the University of Oxford, are assessing its integration into primary care networks [8].
Barriers to Adoption:
Despite its promise, widespread use, particularly in Africa, faces hurdles:
- Advanced diagnostic technology requires significant capital investment and per-test cartridge costs, which are barriers in low-resource settings.
- The system requires regulatory approvals such as; NABL (National Accreditation Board for Testing and Calibration Laboratories), ISO (International Organization for Standardization), or local FDA (Food and Drug Administration).
- Currently, the system is validated only for uncomplicated UTIs and five specific antibiotics. Expanding its panel to include other infection types (e.g., blood or respiratory) and more drugs is necessary for broader utility [7].
The Future Outlook
The PA-100 AST System proves that rapid, point-of-care phenotypic testing is feasible. The future lies in adapting this nanofluidic technology for other bodily fluids (blood, sputum) to combat sepsis and pneumonia and expanding the antibiotic panels to address multi-drug-resistant organisms.
For Africa, the future of this technology raises urgent questions about equity and access. A 2020 systematic review of low- and middle-income countries found that, on average, 52% of patients attending primary care facilities received an antibiotic prescription [10]. The PA-100 could decentralize diagnostics to rural clinics and preserve affordable antibiotics by ensuring they remain effective.
However, major barriers remain, such as; device and cartridge costs and unreliable electricity. The question for African health leaders is whether deliberate investment will make it accessible. Without strategic action, the PA-100 risks becoming another innovation that benefits wealthy nations while bypassing those who need it most.
References
- Yee, R., Dien Bard, J., & Simner, P. J. (2021). The genotype-to-phenotype dilemma: How should laboratories approach discordant susceptibility results? Journal of Clinical Microbiology, 59(6), Article e00138-20. https://journals.asm.org/doi/10.1128/jcm.00138-20
- Sysmex Europe. (2024, September 9). Fundació Puigvert validates a new tool capable of selecting the most suitable antibiotic for uncomplicated urinary tract infections within 45 minutes. https://www.sysmex-europe.com/company/news-and-events/news-listings/news-details/fundacio-puigvert-validates-a-new-tool-capable-of-selecting-the-most-suitable-antibiotic-for-uncomplicated-urinary-tract-infections-within-45-minutes/
- World Health Organization. (2023, November 17). Antimicrobial resistance. https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance
- Alonso-Tarrés, C., Benjumea Moreno, C., Navarro, F., et al. (2024). Bacteriuria and phenotypic antimicrobial susceptibility testing in 45 min by point-of-care Sysmex PA-100 System: first clinical evaluation. European Journal of Clinical Microbiology & Infectious Diseases, 43, 1533–1543. https://pmc.ncbi.nlm.nih.gov/articles/PMC11271345/
- Sysmex Suisse AG. (n.d.). PA-100 AST System. Retrieved on April 22, 2026, from https://www.sysmex.ch/produkte/products-detail/pa-100-ast-system/
- Science Museum Group. (n.d.). PA-100 AST System main unit. Science Museum Group Collection. https://collection.sciencemuseumgroup.org.uk/objects/co9018903/pa-100-ast-system-main-unit
- PR Newswire. (2024, June 12). From 3 days to 45 minutes: Rapid test for UTI wins £10M Longitude Prize on AMR to transform fight against superbugs. https://www.prnewswire.com/news-releases/from-3-days-to-45-minutes-rapid-test-for-uti-wins-10m-longitude-prize-on-amr-to-transform-fight-against-superbugs-302169734.html
- The Pharmaceutical Journal. (2024, June 12). Rapid UTI test wins £8m Longitude Prize on antimicrobial resistance. https://pharmaceutical-journal.com/article/news/rapid-uti-test-wins-8m-longitude-prize-on-antimicrobial-resistance
- Longitude Prize on AMR. (n.d.). About the prize. https://amr.longitudeprize.org/about-the-prize/
- Sulis, G., Adam, P., Nafade, V., Gore, G., Daniels, B., Daftary, A., Das, J., Gandra, S., & Pai, M. (2020). Antibiotic prescription practices in primary care in low- and middle-income countries: A systematic review and meta-analysis. PLOS Medicine, 17(6), e1003139. https://journals.plos.org/plosmedicine/article?id=10.1371/journal.pmed.1003139

